The lack of a clinically viable method to track and direct cancer drugs to tumors is a big problem for targeted therapeutics.
But a new ultrasonic method proposed by biomedical engineers from Qifa Zhou’s team at the University of Southern California in Los Angeles could enable acoustic control and real-time tracking of drug release within the body. The researchers report on their manipulation of ultrasonic waves to pinpoint drug delivery in Applied Physics Letters, from AIP Publishing.
“In conventional drug delivery, tissue is examined ex vivo under the microscope, or radioactive materials are used to trace drugs in vivo. We propose a new way to image and move the drug precisely inside the human body by combining the new plane wave imaging method with a focused ultrasound transducer,” said post-doctoral researcher Xuejun Qian.
Accurate drug delivery is crucial to ensure tumor obliteration, while avoiding the toxic side effects of cancer therapeutics on healthy tissue. Ultrasound is a popular method for noninvasively imaging inside the body. But because the conventional method lacks sensitivity, it has not been used in drug delivery previously. Zhou’s team adapted a new, ultrafast ultrasound method that eliminates background noise to accurately track a drug delivery vehicle within a phantom blood vessel.
[ad_336]
Hanmin Peng, a visiting scholar from Nanjing University of Aeronautics & Astronautics, China, and co-workers pumped water through a narrow silicone tube to mimic blood flow through a blood vessel. They placed the tube beneath real pig tissue and imaged across this to make the setup more realistic. Microbubbles, tiny pockets of air, that can be used as vehicles for drug delivery were introduced into the fake blood vessels.
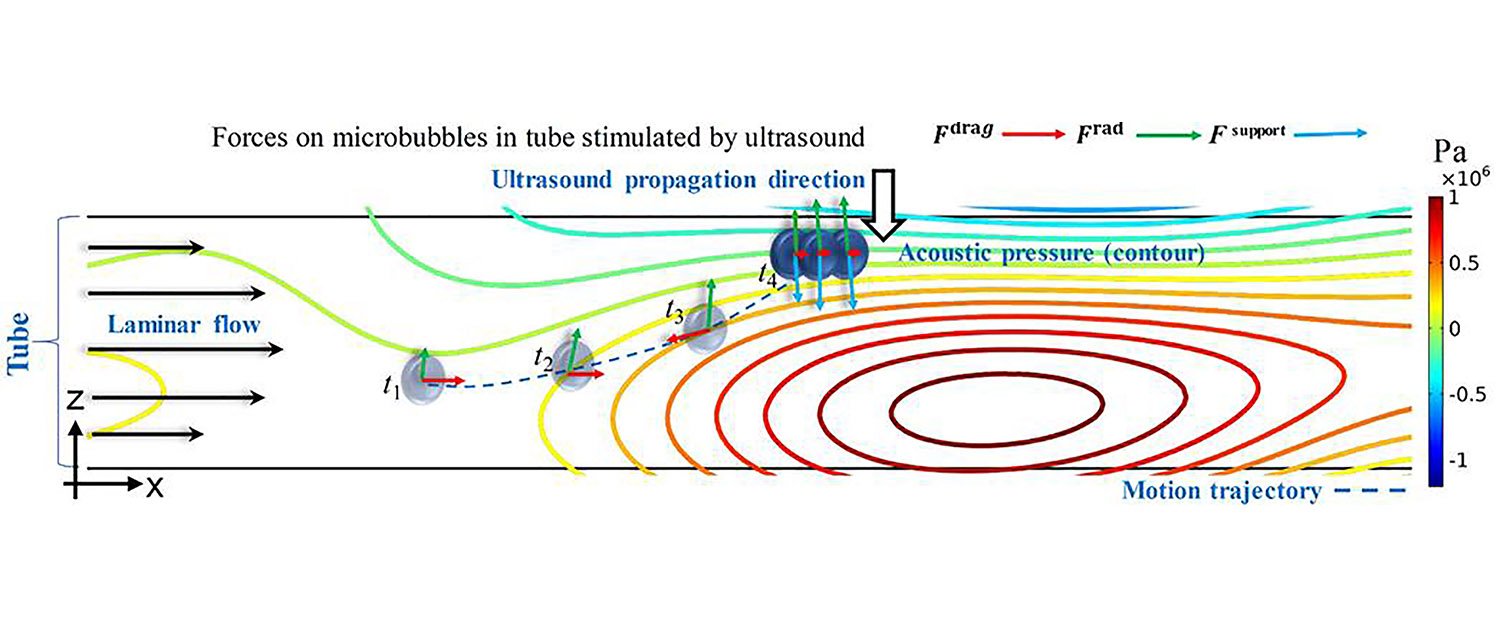
In recent years, there’s been much excitement over the ability to focus sound waves into “acoustic tweezers,” which can manipulate particles. Zhou’s team applied a focused ultrasound transducer to trap the microbubbles identified by their ultrafast imaging system.
The team predicted microbubble motion and calculated the acoustic radiation forces required to trap and move the bubbles to specific areas in the phantom blood vessel.
By balancing the acoustic radiation force from the transducer, the team moved the trapped microbubbles to a specific location on the tube wall and turned up the acoustic power to burst the bubbles.
[rand_post]
Ultrasound waves vibrate the air contained within microbubbles, which enabled Peng and co-workers to use their novel ultrafast ultrasound imaging system to precisely track the microbubbles at depths of up to 10 millimeters within the tissue. They hope this combination of ultrasound tracking and targeting can be translated to noninvasively directing drug-containing microbubbles to blood vessels adjacent to tumor locations in the body.
“We want to try in vivo studies on rat or rabbit to see whether the proposed method can monitor and release microbubble-based drug delivery in a real body,” said Qian. “We hope to further improve the imaging resolution, sensitivity and speed within a real case, and if it works, the long-term goal would be to move towards a human study.”